1. Photon of frequency $$\nu $$ has a momentum associated with it. If $$c$$ is the velocity of light, the momentum is
A.
$$\frac{{h\nu }}{c}$$
B.
$$\frac{\nu }{c}$$
C.
$$h\nu c$$
D.
$$\frac{{h\nu }}{{{c^2}}}$$
Answer :
$$\frac{{h\nu }}{c}$$
2. A telephonic communication service is working at carrier frequency of $$10\,GHz.$$ Only $$10\% $$ of it is utilized for transmission. How many telephonic channels can be transmitted simultaneously if each channel requires a bandwidth of $$5\,kHz$$ ?
A.
$$2 \times {10^3}$$
B.
$$2 \times {10^4}$$
C.
$$2 \times {10^5}$$
D.
$$2 \times {10^6}$$
Answer :
$$2 \times {10^5}$$
3.
In a historical experiment to determine Planck's constant, a metal surface was irradiated with light of different wavelengths. The emitted photoelectron energies were measured by applying a stopping potential. The relevant data for the wavelength $$\left( \lambda \right)$$ of incident light and the corresponding stopping potential $$\left( {{V_0}} \right)$$ are given below :
$$\lambda \left( {\mu m} \right)$$
$${V_0}$$ (Volt)
0.3
2.0
0.4
1.0
0.5
0.4
Given that $$c = 3 \times {10^8}m{s^{ - 1}}$$ and $$e = 1.6 \times {10^{ - 19}}C,$$ Planck's constant (in units of $$Js$$ ) found from such an experiment is
A.
$$6.0 \times {10^{ - 34}}$$
B.
$$6.4 \times {10^{ - 34}}$$
C.
$$6.6 \times {10^{ - 34}}$$
D.
$$6.8 \times {10^{ - 34}}$$
Answer :
$$6.4 \times {10^{ - 34}}$$
4.
The surface of a metal is illuminted with the light of $$400\,nm.$$ The kinetic energy of the ejected photo electrons was found to be $$1.68\,eV.$$ The work function of the metal is :
$$\left( {hc = 1240\,eV.nm} \right)$$
A.
$$1.41\,eV$$
B.
$$1.51\,eV$$
C.
$$1.68\,eV$$
D.
$$3.09\,eV$$
Answer :
$$1.41\,eV$$
5. During the propagation of electromagnetic waves in a medium:
A.
Electric energy density is double of the magnetic energy
density.
B.
Electric energy density is half of the magnetic energy
density.
C.
Electric energy density is equal to the magnetic energy density.
D.
Both electric and magnetic energy densities are zero.
Answer :
Electric energy density is equal to the magnetic energy density.
6. Radiation of wavelength $$\lambda ,$$ is incident on a photocell. The fastest emitted electron has speed $$\nu .$$ If the wavelength is changed to $$\frac{{3\lambda }}{4},$$ the speed of the fastest emitted electron will be:
A.
$$ = \nu {\left( {\frac{4}{3}} \right)^{\frac{1}{2}}}$$
B.
$$ = \nu {\left( {\frac{3}{4}} \right)^{\frac{1}{2}}}$$
C.
$$ > \nu {\left( {\frac{4}{3}} \right)^{\frac{1}{2}}}$$
D.
$$ < \nu {\left( {\frac{4}{3}} \right)^{\frac{1}{2}}}$$
Answer :
$$ > \nu {\left( {\frac{4}{3}} \right)^{\frac{1}{2}}}$$
7. The anode voltage of a photocell is kept fixed. The wavelength $$\lambda $$ of the light falling on the cathode is gradually changed. The plate current $$I$$ of the photocell varies as follows :
A.
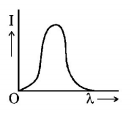

B.
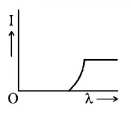

C.
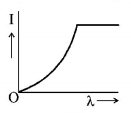

D.
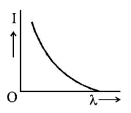

Answer :


8. $${K_\alpha }$$ wavelength emitted by an atom of atomic number $$Z = 11$$ is $$\lambda .$$ Find the atomic number for an atom that emits $${K_\alpha }$$ radiation with wavelength $$4\lambda .$$
A.
$$Z = 6$$
B.
$$Z = 4$$
C.
$$Z = 11$$
D.
$$Z = 44$$
Answer :
$$Z = 6$$
9. The work function of a substance is $$4.0\,eV.$$ The longest wavelength of light that can cause photoelectron emission from this substance is approximately.
A.
$$310\,nm$$
B.
$$400\,nm$$
C.
$$540\,nm$$
D.
$$220\,nm$$
Answer :
$$310\,nm$$
10.
This question has Statement - 1 and Statement - 2. Of the four choices given after the statements, choose the one that best describes the two statements.
Statement - 1: A metallic surface is irradiated by a monochromatic light of frequency $$v > {v_0}$$ (the threshold frequency). The maximum kinetic energy and the stopping potential are $${K_{\max }}$$ and $${V_0}$$ respectively. If the frequency
incident on the surface is doubled, both the $${K_{\max }}$$ and $${V_0}$$ are also doubled.
Statement - 2 : The maximum kinetic energy and the stopping potential of photoelectrons emitted from a surface are linearly dependent on the frequency of incident light.
A.
Statement-1 is true, Statement-2 is true, Statement -2 is the correct explanation of Statement - 1.
B.
Statement-1 is true, Statement-2 is true, Statement-2 is not the correct explanation of Statement-1.
C.
Statement-1 is false, Statement-2 is true.
D.
Statement-1 is true, Statement-2 is false.
Answer :
Statement-1 is false, Statement-2 is true.