1. Cyclohexylamine is stronger base than aniline because
A.
in aniline electron pair is involved in conjugation
B.
in cyclohexylamine electron pair is involved in conjugation
C.
in aniline $$ - N{H_2}$$ group is protonated
D.
in cyclohexylamine nitrogen has a negative charge
Answer :
in aniline electron pair is involved in conjugation
2. The order of basicity of amines in gaseous state is :
A.
\[{{1}^{\circ }}>{{2}^{\circ }}>{{3}^{\circ }}>N{{H}_{3}}\]
B.
\[{{3}^{\circ }}>{{2}^{\circ }}>N{{H}_{3}}>{{1}^{\circ }}\]
C.
\[{{3}^{\circ }}>{{2}^{\circ }}>{{1}^{\circ }}>N{{H}_{3}}\]
D.
\[N{{H}_{3}}>{{1}^{\circ }}>{{2}^{\circ }}>{{3}^{\circ }}\]
Answer :
\[{{3}^{\circ }}>{{2}^{\circ }}>{{1}^{\circ }}>N{{H}_{3}}\]
3. Indicate which nitrogen compound amongst the following would undergo Hofmann reaction?
A.
$$RCONHC{H_3}$$
B.
$$RCOON{H_4}$$
C.
$$RCON{H_2}$$
D.
$$RCONHOH$$
Answer :
$$RCON{H_2}$$
4.
Coupling of diazonium salts of following takes place in the order
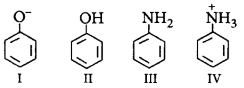
A.
IV < II < III < I
B.
IV > III < II < I
C.
II < IV < I < III
D.
I < II < III < IV
Answer :
IV < II < III < I
5. In the Hofmann bromamide degradation reaction, the number of moles of $$NaOH$$ and $$B{r_2}$$ used per mole of amine produced are:
A.
Two moles of $$NaOH$$ and two moles of $$B{r_2}$$
B.
Four moles of $$NaOH$$ and one mole of $$B{r_2}$$
C.
One mole of $$NaOH$$ and one mole of $$B{r_2}$$
D.
Four moles of $$NaOH$$ and two moles of $$B{r_2}$$
Answer :
Four moles of $$NaOH$$ and one mole of $$B{r_2}$$
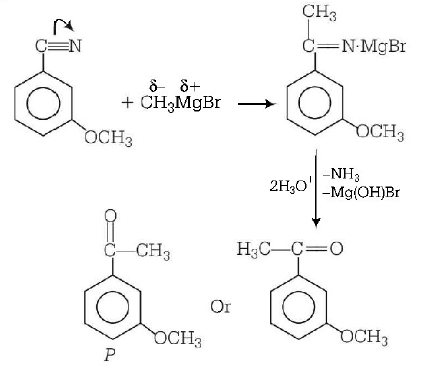
6.
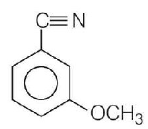 $$ + C{H_3}MgBr \to P$$
$$ + C{H_3}MgBr \to P$$
Product $$P$$ in the above reaction is
A.
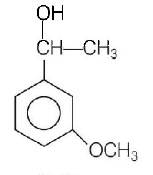

B.
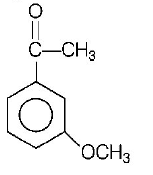

C.
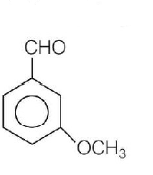

D.
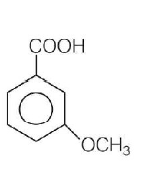

Answer :


7.
In the chemical reactions,
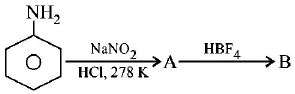
the compounds $$'A'$$ and $$'B'$$ respectively are
A.
nitrobenzene and fluorobenzene
B.
phenol and benzene
C.
benzene diazonium chloride and fluorobenzene
D.
nitrobenzene and chlorobenzene
Answer :
benzene diazonium chloride and fluorobenzene
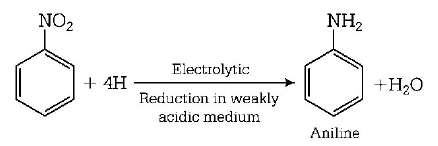
8. Electrolytic reduction of nitrobenzene in weakly acidic medium gives
A.
aniline
B.
nitrosobenzene
C.
$$N$$ - phenyl hydroxylamine
D.
$$p$$ - hydroxyaniline
Answer :
aniline
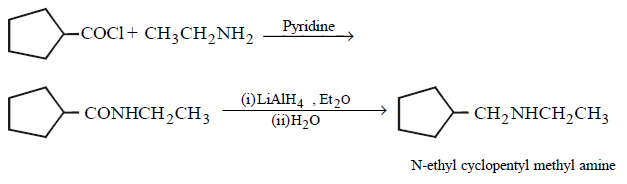
9. Which of the following does not give $$N–$$ ethyl cyclopentylamine as major product ?
A.
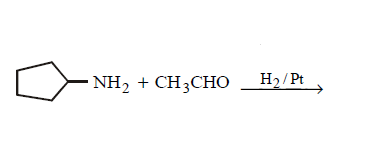
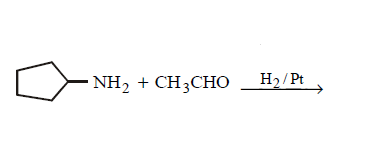
B.
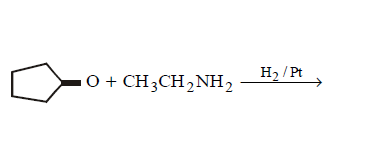
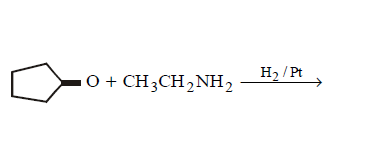
C.
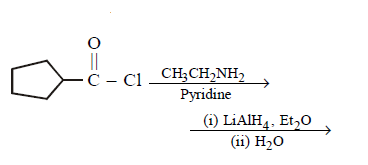

D.
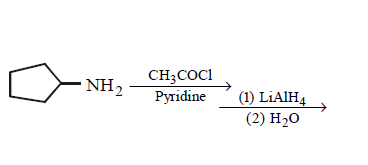

Answer :


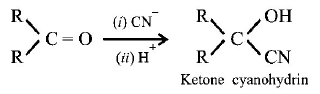
10. The formation of cyanohydrin from a ketone is an example of :
A.
Electrophilic addition
B.
Nucleophilic addition
C.
Nucleophilic substitution
D.
Electrophilic substiution
Answer :
Nucleophilic addition