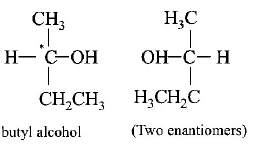
1.
Given below are the structures of few compounds with molecular formula $${C_4}{H_{10}}O.$$ Select metamers from these structures.
\[\begin{align}
& \left( \text{i} \right)C{{H}_{3}}-O-C{{H}_{2}}C{{H}_{2}}C{{H}_{3}} \\
& \left( \text{ii} \right)C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}C{{H}_{2}}OH \\
& \left( \text{iii} \right)C{{H}_{3}}-C{{H}_{2}}-O-C{{H}_{2}}-C{{H}_{3}} \\
& \left( \text{iv} \right)C{{H}_{3}}\underset{\begin{smallmatrix}
|\,\,\,\,\, \\
OH\,\,
\end{smallmatrix}}{\mathop{-CH-}}\,C{{H}_{2}}-C{{H}_{3}} \\
\end{align}\]
A.
(i) and (ii)
B.
(ii) and (iii)
C.
(i) and (iii)
D.
(ii) and (iv)
Answer :
(i) and (iii)
2.
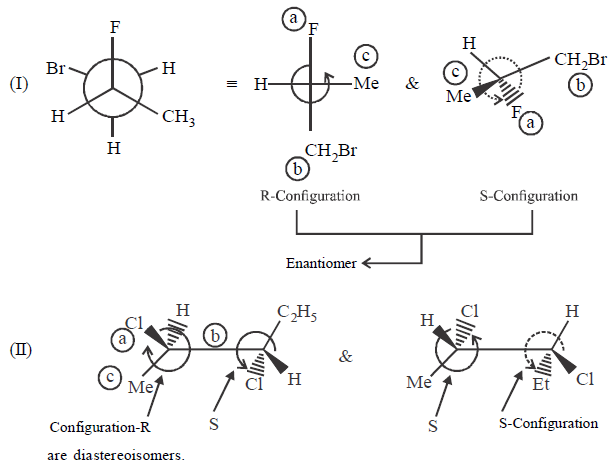
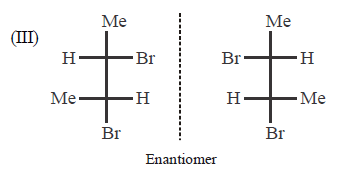
Indicate whether each of the following pairs are identical or not ?
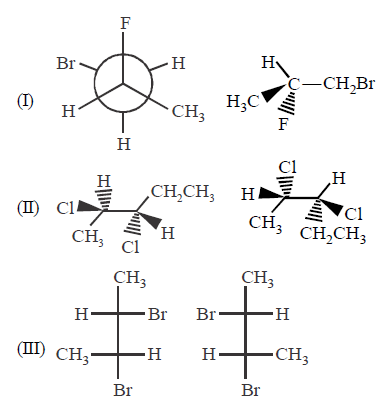
A.
I - enantiomers; II - diastereomers; III - enantiomers
B.
I - identical; II - enantiomers; III - enantiomers
C.
I - enantiomers; II - diastereomers; III - identical
D.
I - enantiomers; II - identical; III - identical
Answer :
I - enantiomers; II - diastereomers; III - enantiomers
3. Which of the following compounds will exhibit geometrical isomerism ?
A.
1 - Phenyl - 2 - butene
B.
3 - Phenyl - 1 - butene
C.
2 - Phenyl - 1 - butene
D.
1,1 - Diphenyl - 1 - propene
Answer :
1 - Phenyl - 2 - butene
4. Allyl isocyanide has
A.
$$9\,\sigma \,\,{\text{and}}\,\,4\,\pi {\text{ - bonds}}$$
B.
$$8\,\sigma \,\,{\text{and}}\,\,5\,\pi {\text{ - bonds}}$$
C.
$$9\,\sigma ,3\,\,\pi \,\,{\text{and}}\,\,2\,\,{\text{non - bonded electrons}}$$
D.
$$8\,\sigma ,3\,\pi \,\,{\text{and}}\,\,4\,\,{\text{non - bonded electrons}}$$
Answer :
$$9\,\sigma ,3\,\,\pi \,\,{\text{and}}\,\,2\,\,{\text{non - bonded electrons}}$$
5. Which one of the following acids does not exhibit optical isomerism ?
A.
Lactic acid
B.
Tartaric acid
C.
Maleic acid
D.
$$\alpha $$ - amino acids
Answer :
Maleic acid
6. Racemic mixture is formed by mixing two
A.
isomeric compounds
B.
chiral compounds
C.
meso compounds
D.
enantiomers with chiral carbon.
Answer :
enantiomers with chiral carbon.
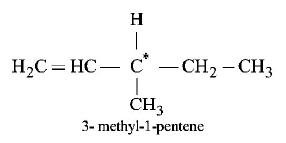
7. Out of the following, the alkene that exhibits optical isomerism is
A.
3 - methyl - 2 - pentene
B.
4 - methyl - 1 - pentene
C.
3 - methyl - 1 - pentene
D.
2 - methyl - 2 - pentene
Answer :
3 - methyl - 1 - pentene
8.
Following types of compounds ( as I, II )
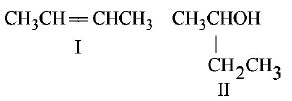
are studied in terms of isomerism in :
A.
chain isomerism
B.
position isomerism
C.
conformers
D.
stereoisomerism
Answer :
stereoisomerism
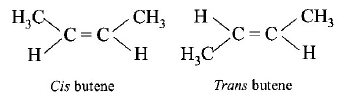
9. Maleic acid and fumaric acids are
A.
Chain isomers
B.
Functional isomers
C.
Tautomers
D.
Geometrical isomers
Answer :
Geometrical isomers
10.
The structure 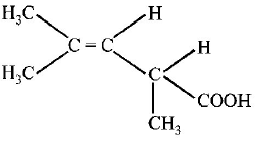 shows :
shows :
A.
geometrical isomersism
B.
optical isomerism
C.
geometrical & optical isomerism
D.
tautomerism.
Answer :
optical isomerism