1.
$$C{H_2}$$  \[C{{H}_{2}}+HBr\xrightarrow{\text{peroxide}}A,\] then $$A$$ is
\[C{{H}_{2}}+HBr\xrightarrow{\text{peroxide}}A,\] then $$A$$ is
A.

B.

C.
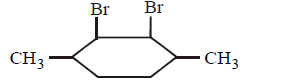

D.
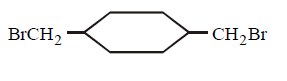

Answer :


2. The negative part of the addendum ( the molecule to be added ) adds on to the carbon atom of the double bond containing the least number of hydrogen atoms. This rule is known as
A.
Saytzeff's rule
B.
Peroxide rule
C.
Markovnikov's rule
D.
Hoffmann rule
Answer :
Markovnikov's rule
3.
Classify the following compounds as primary, secondary and tertiary halides.
(i) 1-Bromobut-2-ene
(ii) 4-Bromopent-2-ene
(iii) 2-Bromo-2-methylpropane
A.
(i) - secondary, (ii) - tertiary, (iii) - primary
B.
(i) - secondary, (ii) - primary, (iii) - tertiary
C.
(i) - primary, (ii) - tertiary, (iii) - secondary
D.
(i) - primary, (ii) - secondary, (iii) - tertiary
Answer :
(i) - primary, (ii) - secondary, (iii) - tertiary
4.
Identify the products $$(A)$$ and $$(B)$$ in the reactions.
$$\eqalign{
& RX + AgCN \to \underline {\,\,\left( A \right)\,\,} + AgX \cr
& RX + KCN \to \underline {\,\,\left( B \right)\,\,} + KX \cr} $$
A.
$$\left( A \right){\text{ - }}RCN,\,\left( B \right){\text{ - }}RCN$$
B.
$$\left( A \right){\text{ - }}RCN,\,\left( B \right){\text{ - }}RNC$$
C.
$$\left( A \right){\text{ - }}RNC,\,\left( B \right){\text{ - }}RCN$$
D.
$$\left( A \right){\text{ - }}RNC,\,\left( B \right){\text{ - }}RNC$$
Answer :
$$\left( A \right){\text{ - }}RNC,\,\left( B \right){\text{ - }}RCN$$
5.
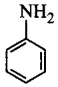 \[\xrightarrow[280\,K]{NaN{{O}_{2}}+HCl}K\xrightarrow{KI}Y\]
\[\xrightarrow[280\,K]{NaN{{O}_{2}}+HCl}K\xrightarrow{KI}Y\]
$$X$$ and $$Y$$ in the reaction are
A.
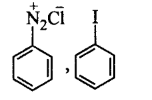

B.
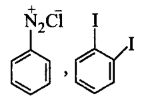

C.
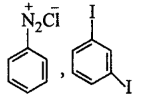

D.
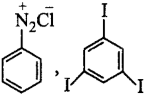

Answer :


6. The position of $$ - Br$$ in the compound $$C{H_3}CH = CHC\left( {Br} \right){\left( {C{H_3}} \right)_2}$$ can be classified as _________.
A.
allyl
B.
aryl
C.
vinyl
D.
secondary
Answer :
allyl
7.
In the following reaction sequence :
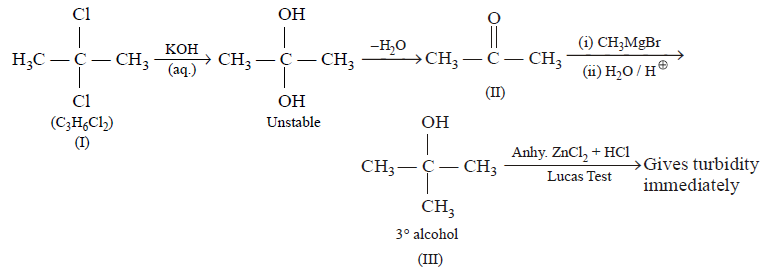
\[\underset{\left( {{C}_{3}}{{H}_{6}}C{{l}_{2}} \right)}{\mathop{I}}\,\xrightarrow{KOH\left( aq \right)}II\xrightarrow[\left( ii \right)\,{{H}_{2}}O/{{H}^{+}}]{\left( i \right)\,C{{H}_{3}}MgBr}III\xrightarrow{Anhy.ZnC{{l}_{2}}+Conc.HCl}\text{given}\,\text{turbidity immediately}\]
The compound $$I$$ is :
A.
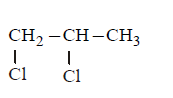
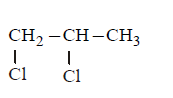
B.
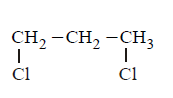
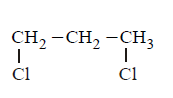
C.
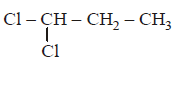
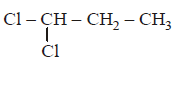
D.
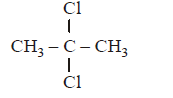
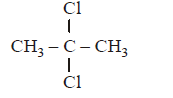
Answer :
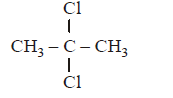
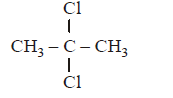
8. Which alkyl halide exhibits complete racemisation in $${S_N}1$$ reaction?
A.
$${\left( {C{H_3}} \right)_2}CHCl$$
B.
$$C{H_3}C{H_2}C{H_2}Cl$$
C.
$$C{H_3}C{H_2}Cl$$
D.
$${C_6}{H_5}C{H_2}Cl$$
Answer :
$${C_6}{H_5}C{H_2}Cl$$
9.
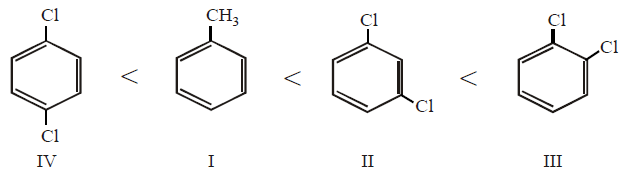
Arrange the following compounds in order of increasing dipole moment :
(I) Toluene
(II) $$m$$ - dichlorobenzene
(III) $$o$$ - dichlorobenzene
(IV) $$p$$ - dichlorobenzene
A.
I < IV < II < III
B.
IV < I < II < III
C.
IV < I < III < II
D.
IV < II < I < III
Answer :
IV < I < II < III
10. A primary alkyl halide would prefer to undergo __________.
A.
$${S_N}1$$ reaction
B.
$${S_N}2$$ reaction
C.
$$\alpha $$ - elimination
D.
racemisation
Answer :
$${S_N}2$$ reaction