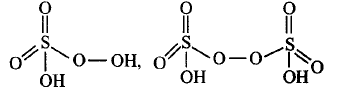
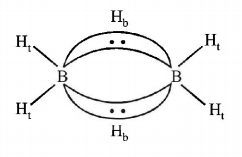
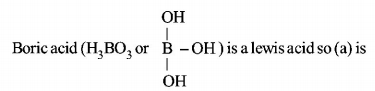1. Which one of the following is the correct statement?
A.
Boric acid is a protonic acid
B.
Beryllium exhibits coordination number of six
C.
Chlorides of both beryllium and aluminium have bridged
chloride structures in solid phase
D.
$${B_2}{H_6}2N{H_3}$$ is known as ‘inorganic benzene’
Answer :
Chlorides of both beryllium and aluminium have bridged
chloride structures in solid phase
2. Which of the following are peroxoacids of sulphur?
A.
$${H_2}S{O_5}\,\,{\text{and}}\,\,{H_2}{S_2}{O_8}$$
B.
$${H_2}S{O_5}\,\,{\text{and}}\,\,{H_2}{S_2}{O_7}$$
C.
$${H_2}{S_2}{O_7}\,\,{\text{and}}\,\,{H_2}{S_2}{O_8}$$
D.
$${H_2}{S_2}{O_6}\,\,{\text{and}}\,\,{H_2}{S_2}{O_7}$$
Answer :
$${H_2}S{O_5}\,\,{\text{and}}\,\,{H_2}{S_2}{O_8}$$
3. Which of the following bonds will be most polar?
A.
$$N—Cl$$
B.
$$O—F$$
C.
$$N—F$$
D.
$$N—N$$
Answer :
$$N—F$$
4. Bauxite ore is generally contaminated with impurity of oxides of two elements $$X$$ and $$Y.$$ Which of the following statement is correct ?
A.
$$X$$ is a non-metal and belongs to the third period while $$Y$$ is a metal and belongs to the fourth period.
B.
One of two oxides has three-dimensional polymeric structure.
C.
Both (A) and (B) are correct.
D.
None of the above.
Answer :
Both (A) and (B) are correct.
5. $$AlC{l_3}$$ achieves stability by forming a dimer. In trivalent state the compound is hydrolysed in water. $$AlC{l_3}$$ in acidified aqueous solution forms
A.
$$Al{\left( {OH} \right)_3} + HCl$$
B.
$${\left[ {Al{{\left( {{H_2}O} \right)}_6}} \right]^{3 + }} + 3C{l^ - }$$
C.
$$AlC{l_3} \cdot 2{H_2}O$$
D.
$$A{l_2}{O_3} + HCl$$
Answer :
$${\left[ {Al{{\left( {{H_2}O} \right)}_6}} \right]^{3 + }} + 3C{l^ - }$$
6.
In the reaction
$$HN{O_3} + HF \to {H_2}\mathop N\limits^ + {O_3} + {F^ - }$$ base is
A.
$$HF$$
B.
$$HN{O_3}$$
C.
$$HF\,\,{\text{and}}\,\,HN{O_3}$$
D.
$${\text{None of these}}$$
Answer :
$$HN{O_3}$$
7. The brown ring test for $$NO_2^ - $$ and $$NO_3^ - $$ is due to the formation of complex ion with a formula
A.
$${\left[ {Fe{{\left( {{H_2}O} \right)}_6}} \right]^{2 + }}$$
B.
$${\left[ {Fe\left( {NO} \right){{\left( {CN} \right)}_5}} \right]^{2 + }}$$
C.
$${\left[ {Fe{{\left( {{H_2}O} \right)}_5}NO} \right]^{2 + }}$$
D.
$${\left[ {Fe\left( {{H_2}O} \right){{\left( {NO} \right)}_5}} \right]^{2 + }}$$
Answer :
$${\left[ {Fe{{\left( {{H_2}O} \right)}_5}NO} \right]^{2 + }}$$
8.
From the following information
$$X + {H_2}S{O_4} \to Y$$ ( a colourless and irritating gas )
$$Y + {K_2}C{r_2}{O_7} + {H_2}S{O_4} \to $$ ( green coloured solution )
Identify the pair $$X$$ and $$Y.$$
A.
$$\bar Cr,HCl$$
B.
$$SO_3^{2 - },S{O_2}$$
C.
$${S^{2 - }},{H_2}S$$
D.
$$CO_3^{2 - },C{O_2}$$
Answer :
$$SO_3^{2 - },S{O_2}$$
9. The structure of diborane $$\left( {{B_2}{H_6}} \right)$$ contains
A.
four $$2c - 2e$$ bonds and four $$3c - 2e$$ bonds
B.
two $$2c - 2e$$ bonds and two $$3c - 3e$$ bonds
C.
two $$2c - 2e$$ bonds and four $$3c - 2e$$ bonds
D.
four $$2c - 2e$$ bonds and two $$3c - 2e$$ bonds
Answer :
four $$2c - 2e$$ bonds and two $$3c - 2e$$ bonds
10.
What is the correct observation when $$B{r_2}$$ is treated with $$NaF, NaCl$$ and $$NaI$$ taken in three test-tubes labelled as $$(X), (Y)$$ and $$(Z)?$$
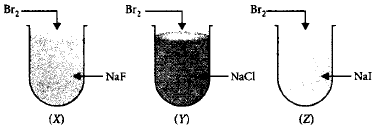
A.
$${F_2}$$ is liberated in $$(X)$$ and $$C{l_2}$$ in $$(Y).$$
B.
Only $${I_2}$$ is liberated in $$(Z).$$
C.
Only $$C{l_2}$$ is liberated in $$(Y).$$
D.
Only $${F_2}$$ is liberated in $$(X).$$
Answer :
Only $${I_2}$$ is liberated in $$(Z).$$