1.
During hydrate formation from aqueous solution, water can be associated in different forms. Indicate the wrong combination.
(i) Coordinated water - $${\left[ {Cr{{\left( {{H_2}O} \right)}_6}} \right]^{3 + }}3C{l^ - }$$
(ii) Interstitial water - $$BaC{l_2} \cdot 2{H_2}O$$
(iii) Hydrogen bonded water - $${\left[ {Cu{{\left( {{H_2}O} \right)}_4}} \right]^{2 + }}SO_4^{2 - } \cdot {H_2}O$$
A.
(i)
B.
(ii)
C.
(iii)
D.
None of these
Answer :
None of these
2. The production of dihydrogen obtained from coal gasification can be increased by reacting carbon monoxide of syngas mixture with steam in presence of a catalyst iron chromate. What is this process called?
A.
Hydrogen reaction
B.
Water-gas shift reaction
C.
Coal-gas shift reaction
D.
Syn gasification
Answer :
Water-gas shift reaction
3. In complex hydrides, hydride ions act as ligand and are coordinated to metal ions. These hydrides are good reducing agents. Which of the following hydrides is not a complex hydride?
A.
$$LiAl{H_4}$$
B.
$$NaB{H_4}$$
C.
$${\left( {Al{H_3}} \right)_n}$$
D.
$$LiB{H_4}$$
Answer :
$${\left( {Al{H_3}} \right)_n}$$
4. When same amount of zinc is treated separately with excess of sulphuric acid and excess of sodium hydroxide solution the ratio of volumes of hydrogen evolved is
A.
1 : 1
B.
1 : 2
C.
2 : 1
D.
9 : 4
Answer :
1 : 1
5. Which of the following reactions is not used in preparation of deuterium compounds using heavy water?
A.
$$Ca{C_2} + 2{D_2}O \to {C_2}{D_2} + Ca{\left( {OD} \right)_2}$$
B.
$$S{O_3} + {D_2}O \to {D_2}S{O_4}$$
C.
$$2AlN + 3{D_2}O \to A{l_2}{O_3} + 2ND$$
D.
$$A{l_4}{C_3} + 12{D_2}O \to 3C{D_4} + 4Al{\left( {OD} \right)_3}$$
Answer :
$$2AlN + 3{D_2}O \to A{l_2}{O_3} + 2ND$$
6. A commercial sample of hydrogen peroxide is labelled as 10 volume. Its percentage strength is nearly
A.
3%
B.
1%
C.
90%
D.
10%
Answer :
3%
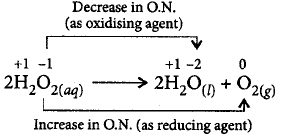
7. Hydrogen peroxide is __________.
A.
an oxidising agent
B.
a reducing agent
C.
both an oxidising and a reducing agent
D.
neither oxidising nor reducing agent
Answer :
both an oxidising and a reducing agent
8. Permanent hardness in water cannot be cured by :
A.
Treatment with washing soda
B.
Boiling
C.
Calgon’s method
D.
Ion exchange method
Answer :
Boiling
9. Some of the major uses of heavy water are given below. Which one is not correct?
A.
It is used as a moderator in nuclear reactors.
B.
It is used as a tracer compound for studying reaction mechanism.
C.
High concentration of heavy water accelerates the growth of plants.
D.
It is used in preparing deuterium.
Answer :
High concentration of heavy water accelerates the growth of plants.
10. If a mole of hydrogen molecule is heated to a high temperature then which of the following reactions take place?
A.
$${H_{2\left( g \right)}} + 436\,kJ\,mo{l^{ - 1}} \to {H_{\left( g \right)}} + {H_{\left( g \right)}}$$
B.
$$2{H_{2\left( g \right)}} + 820\,kJ\,mo{l^{ - 1}} \to 2{H_{2\left( l \right)}}$$
C.
$${H_{2\left( g \right)}} + {H_{2\left( g \right)}} + 436\,kJ\,mo{l^{ - 1}} \to $$ $$H_{\left( {aq} \right)}^ + + H_{\left( {aq} \right)}^ - $$
D.
$${H_{2\left( g \right)}} + 200\,kJ\,mo{l^{ - 1}} \to {H_{\left( g \right)}} + {H_{\left( g \right)}}$$
Answer :
$${H_{2\left( g \right)}} + 436\,kJ\,mo{l^{ - 1}} \to {H_{\left( g \right)}} + {H_{\left( g \right)}}$$